Free Radicals
 What
are Free Radicals?
What
are Free Radicals?
A free radical is a chemical with a very reactive chemical bond, the chemical bonds which hold atoms together to make molecules contain pairs of electrons. For example there are two electrons in each of the bonds holding the hydrogens to the oxygen in the water molecule. The two electrons stabilise the bond between the atoms. However, some molecules, especially those containing oxygen, can easily gain only one of a pair of these bonding electrons. In a sense we have a molecule with a free chemical bond.
Free radicals are by-products of the natural metabolism in the human body and during the body breaking down foods. There are various external factors cause free radicals to exist in our body such as pollution, pesticides, tobacco smoke, industrial chemicals/cleaners, and solar radiation.
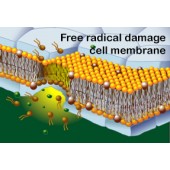
What Makes Free Radicals and Oxidation Harmful?
A free radical is a molecule containing unpaired electrons.
An unpaired electron makes the molecule very reactive. The molecule will
“steal” an electron from other molecules in order to gain a pair for its’ lone
electron. The structure of the molecule that loses the electron is subsequently
changed and can often become a free radical itself.
The most reactive free radical molecule known is the hydroxyl radical. It can be made by X-rays or gamma rays splitting water molecules. The hydroxyl radical, written HO• to show the lone electron, is so reactive that it only takes one billionth of a second to react with a neighbouring molecules, usually by stealing hydrogen atoms from other molecules. The hydroxyl radical regains the lost electron in this manner and forms a water molecule (H2O). To regain its’ electron pair, the hydroxyl radical has changed another molecule by removing a hydrogen electron from it. This causes further reactions leading to a major chemical change in the molecule.
If the hydroxyl radical reacts with our DNA cell, a genetic mutation may occur resulting in a cancer gene being switched on.
Wrinkled skin, stiff joints and hardening of the arteries are general examples of what free radicals will do to us. The damage caused by free radicals accelerates the ageing process. Other conditions that are closely linked to free radical activity include Crohn’s disease, Gallstones, Pancreatitis, Ulcerative colitis, Cataracts, Cancer, Cardiovascular disease, Chronic fatigue syndrome, Rheumatoid arthritis, Alzheimer’s disease, Learning difficulties, Memory problems, Multiple sclerosis, Parkinson’s disease, Emphysema, Male infertility, Hair loss, Skin problems, and possibly many other conditions.
Powered by OpenCart Made by ThemeGlobal - OpenCart Template Club



